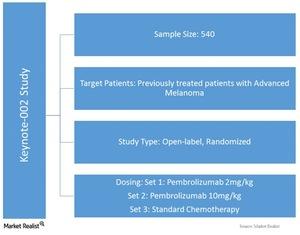 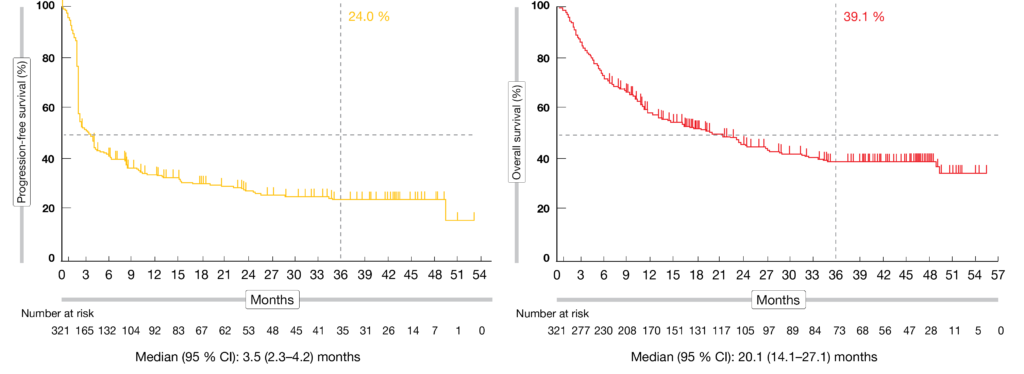
Cassier, MD3 Aurelien Marabelle, MD, PhD4 Aaron R. O'Malley, MD1 Giovanni Mendonca Bariani, MD2 Philippe A. Abstract presented at: Society of Gynecological Oncology 2021 Virtual Annual Meeting on Women’s Cancer March 19-21, 2021 Virtual. Pembrolizumab in Patients With Microsatellite InstabilityHigh Advanced Endometrial Cancer: Results From the KEYNOTE-158 Study David M. Pembrolizumab for Vulvar Squamous Cell Carcinoma: Results From the Phase 2 KEYNOTE-158 Study. As we could see, some of the responses were very durable and therefore meaningful.įrommer RS, Mileshkin L, Manzyuk L, et al. Denis Smith, M.D., Rocio Garcia-Carbonero, M.D., Ph.D., Manuel Benavides, M.D., Ph.D., Peter Gibbs, M.D., Christelle de la Fouchardiere, M.D., Fernando Rivera, M.D., Ph.D. Interestingly, response was irrespective of PD-L1 expression, unlike in cervical cancer, so patients could respond whether they had a PD-L1–positive tumor PD-L1–negative expression. Patients received pembrolizumab 200 mg every three weeks (KEYNOTE-158) or 10 mg/kg every two weeks (KEYNOTE-028) for 2 years. In a 10-cohort phase II KEYNOTE-158 trial of pembrolizumab for previously treated patients with advanced non-CRC tumors, the efficacy of TMB-H (10 mutations/megabase) was assessed as a prospectively planned retrospective analysis. Indeed, low percentage of response, but significant and meaningful responses for some of these patients. Among these responding patients, median duration of response was 20 months. Similar to checkpoint inhibitors, PDAC vaccines have not demonstrated efficacy. One hundred and one patients were treated on the study and 11% responded to the treatment (10.9%) 1 had a complete response and 10 had partial responses. Since then, however, the phase 2 KEYNOTE-158 trial evaluating pembrolizumab in non-colorectal MSI-H/dMMR tumors demonstrated only three partial responses and one complete response out of 22 patients, with an OS of 4 months. Efficacy was evaluated in KEYNOTE-158 (NCT02628067), a multicenter, non-randomized, open-label, multi-cohort trial in 90 patients with unresectable or metastatic MSI-H or dMMR endometrial. According to analyses of the phase II KEYNOTE-158 trial that investigated the efficacy of pembrolizumab monotherapy for patients with solid tumours (n 1073). In a presentation at the Society of Gynecological Oncology (SGO) 2021 Virtual Annual Meeting on Women’s Cancer, Shapira-Frommer presented data showing objective responses were achievable in this patient subgroup with the PD-1 inhibitor. CancerNetwork® sat down with Ronnie Shapira-Frommer, MD, to discuss the antitumor activity of pembrolizumab monotherapy in a cohort of patients with previously treated vulvar squamous cell carcinoma from the phase 2 KEYNOTE-158 trial (NCT02628067).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
